Standards for Produce of Human Consumption
Food Safety Modernisation Act (FSMA): Produce Rule
Standards for the Growing, Harvesting, Packing, and Holding of Produce for Human Consumption
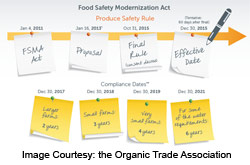 As part of the Food Safety Modernization Act (FSMA), the US Food and Drug Administration (FDA) has issued several rules to clarify regulatory requirements. The rule regarding Standards for the Growing, Harvesting, Packing, and Holding of Produce for Human Consumption was developed after extensive consultation with numerous stakeholders and it follows the same definition for farms that are laid out in the Preventive Controls for Human Food Rule. Here are some of the most important aspects of this rule:
As part of the Food Safety Modernization Act (FSMA), the US Food and Drug Administration (FDA) has issued several rules to clarify regulatory requirements. The rule regarding Standards for the Growing, Harvesting, Packing, and Holding of Produce for Human Consumption was developed after extensive consultation with numerous stakeholders and it follows the same definition for farms that are laid out in the Preventive Controls for Human Food Rule. Here are some of the most important aspects of this rule:
Agricultural Water
No E. coli is allowed in water that may transfer the contamination directly or indirectly to produce. Examples include water that is used:
- for sprout irrigation
- to make ice for storage or
- for washing hands during harvesting.
Water that is applied directly to growing produce (not including sprouts) can have some E. Coli in it, using two measurements: the Geometric Mean (GM), which is an average level for the water source and the Statistical Threshold (STV) which represents the variability of the water source. The FDA intends to have an on-line tool that allows farmers to input their water sample data and calculate these values. The GM must be 126 or less and the STV must be 410 Colony Forming Units (CFU) or less of generic E. Coli per 100 mL of water.
These measurements are meant to be flexible enough to allow for slightly elevated readings due to exceptionally high rains, for example, while maintaining consumer safety. The FDA intends to develop an on-line calculator to help farmers stay in compliance.
If water samples exceed allowable values, corrective action must be taken within a year. This might include treating the water, allowing the microbes to die off between the last irrigation and the harvest, or storing and treating the produce after harvest to remove harmful microbes.
Water from public water systems or that is treated in compliance with the regulations need not be tested.
Biological Soil Amendments
Raw Manure
While the FDA is conducting extensive risk assessments of raw manure usage, the rule suggests that farmers follow the USDA’s National Organic Program standards, which call for a 120-day interval between the application of raw manure for crops in contact with the soil and 90 days for crops not in contact with the soil.
Stabilized Compost
The rule includes two examples of scientifically valid composting methods that meet the standards for minimizing the presence of several harmful agents including Salmonella and listeria as well as E. coli. Even then, as with manure, the compost must be applied in a manner that minimizes the potential for contact with produce during and after application.
Sprouts
Sprouts have been associated with a high number of very serious cases of foodborne illnesses and the new food safety regulations include special requirements for them. These include:
- Treating beans or seeds used for sprouting
- Measures to prevent introduction of dangerous microbes to the soil
- Testing spent irrigation water from each batch of sprouts
- Testing each production batch of the sprouts themselves
- Testing the growing, harvesting and packaging areas
- Taking corrective actions if any of these tests reveal the presence any pathogens.
Domestic and Wild Animals
Animal incursions into a produce growing area represent a contamination risk via fecal matter. If this occurs, farmers must not harvest those plants for human consumption. Farmers are asked to do everything reasonably possible to avoid animal incursions into their fields, but the rule makes it clear that they are not required to destroy animal habitat or clear surrounding areas. They are to visually inspect their crops and clearly mark areas where incursions have occurred.
The rule doesn’t specify times to allow between grazing and harvesting if farmers allow domestic animals to graze in crop fields, but the FDA will provide guidelines later, if needed.
Worker Training, Health and Hygiene
Basic common sense requirements are included here, such as:
- Preventing ill or infected employees from having contact with the produce
- Requiring basic hygiene such as hand washing after bathroom use
- Making sure visitors are not allowed to contaminate the produce
- Giving employees basic hygiene training
Equipment, Tools and Buildings
This part of the rule outlines procedures for ensuring the equipment, tools and work surfaces used for holding, cleaning and packaging produce are clean.
Exemptions
The rule does not apply to:
- Produce that is not consumed as a raw agricultural commodity. The FDA has recognized that most Americans are unlikely to eat the following foods raw:
- asparagus; black beans, great Northern beans, kidney beans, lima beans, navy beans, and pinto beans; garden beets (roots and tops) and sugar beets; cashews; sour cherries; chickpeas; cocoa beans; coffee beans; collards; sweet corn; cranberries; dates; dill (seeds and weed); eggplants; figs; horseradish; hazelnuts; lentils; okra; peanuts; pecans; peppermint; potatoes; pumpkins; winter squash; sweet potatoes; and water chestnuts
- Food grains including barley, dent- or flint-corn, sorghum, oats, rice, rye, wheat, amaranth, quinoa, buckwheat, and oilseeds (e.g. cotton seed, flax seed, rapeseed, etc.)
- Produce grown for personal consumption
- Farms that have sold less than $25,000 worth of produce over the last three years
Modified and/or qualified exemptions are granted to produce that will be further processed in a way that reduces any microorganisms to below acceptable levels, as well as very small or small farms (as defined below), although they must still maintain careful records. The exemption can be withdrawn if an outbreak is traced directly to the farm or if the FDA determines that it is necessary to prevent or mitigate an outbreak. Reinstatement can occur if further investigation shows the farm was not the source of the outbreak or if the FDA is satisfied that the cause has been adequately dealt with.
Variances
Importers into the United States can request variances, if, for example, local growing conditions make adherence too difficult, as long as they can prove that the subject produce will not be adulterated and that there is no more risk to public safety than there would be if they complied with the rule.
The variance must be submitted by a competent authority and must be backed up with scientific data.
Compliance Dates
Compliance dates vary depending on the size of the business and are defined as follows:
- Very small businesses are those which have averaged between $25,000 and $250,000 annual produce sales during the previous three years
- Small businesses are those which have averaged between $250,000 and $500,000
Compliance dates for all but those producing sprouts are:
- Four years for very small businesses
- Three years for small businesses
- Two years for all others
- Also two years’ additional time to comply with the water quality requirements.
Compliance dates for sprout producers are:
- Three years for very small businesses
- Two years for small businesses
- One year for all others
- No additional time allowed to comply with the water quality requirements.
For farms eligible for a qualified exemption, the compliance dates are:
- Immediately for records supporting eligibility for a qualified exemption
- January 1, 2020 for the labeling requirement (if applicable)
- For all other modified requirements:
- Four years for very small businesses
- Three years for small businesses
Environmental Impact Statement
The FDA has published an Environment Impact Statement which places this rule in the context of its likely effect on the environment.
Assistance to Industry
The FDA will make various guidance documents around compliance and modified exemptions available, including those relating to small and very small businesses. Training and technical assistance will be a priority. They have already established the FDA FSMA Food Safety Technical Assistance Network, to provide a central source of information to support industry’s understanding and implementation of the FSMA.
For more information on how meet compliance for the Produce Rule please visit the FDA’s website.

-
 FeaturedRisk management
The Cost of a Breach: What a Cyberattack Could Mean for Food Safety Recalls
FeaturedRisk management
The Cost of a Breach: What a Cyberattack Could Mean for Food Safety Recalls
-
 FeaturedRisk management
Securing the Food Chain: How ISO/IEC 27001 Strengthens Cybersecurity
FeaturedRisk management
Securing the Food Chain: How ISO/IEC 27001 Strengthens Cybersecurity
-
 FeaturedRisk management
Revolutionizing Food Safety Training: Breaking Out of the “Check-the-Box” Mentality
FeaturedRisk management
Revolutionizing Food Safety Training: Breaking Out of the “Check-the-Box” Mentality
-
 GFSI Standards
GFSI 2025: Building Trust, Tech-Forward Solutions, and Global Unity in Food Safety
GFSI Standards
GFSI 2025: Building Trust, Tech-Forward Solutions, and Global Unity in Food Safety
-
 FeaturedFood Safety
Integrated Pest Management: Strategies to Protect Your Brand’s Reputation
FeaturedFood Safety
Integrated Pest Management: Strategies to Protect Your Brand’s Reputation
-
 FeaturedFood Safety Culture & Training
No Open Door Policy: Challenges That Impact Pest Control in Food Processing Plants
FeaturedFood Safety Culture & Training
No Open Door Policy: Challenges That Impact Pest Control in Food Processing Plants



