Food Safety Audits
Audits are a crucial component of maintaining food safety standards and certification, by providing transparency and assurance that standards are being maintained. This transparency increases the collaborative capacity of stakeholders across the supply chain, and enhances safety, efficiency and continual improvement within individual organizations.
Whether you are a seasoned audit participant or new to the game entirely, there is always room for growth and development within your audit process. Auditing does not just have to be just a necessary component of certification or business – it can also be a great opportunity to drive value within your organization.
There are numerous reasons why organizations conduct food safety audits. For instance, auditing is a required component of becoming certified to a standard. Some other reasons to conduct audits include:
- Evaluation of the management system or management priorities
- Commercial objectives
- Supplier evaluations or customer requirements
- Incidents at other facilities
- Regulatory or contractual requirements
Standards-Based Food Safety Audits
With supply chains becoming increasingly globalized and complex, the need for standardized, internationally accepted food safety plans grew. In response to this need, the Global Food Safety Initiative (GFSI), an international committee formed in May 2000 and coordinated by The Consumer Goods Forum, created a system whereby certain food safety schemes are benchmarked against specific criteria. Certification to a GFSI-recognized global standard, such as FSSC 22000, SQF or BRC, gives assurance to customers that specific food safety and quality guidelines are routinely attained, imparting a beneficial stance in the marketplace as a trusted source. Aside from increasing the efficiency of transactions in the supply chain, food safety certification also increases the cost efficiency of the food safety audit process, by allowing businesses to consolidate numerous and costly supplier audits into fewer, standards-based audits.
Businesses may be subjected to numerous proprietary or 2nd party audits each year – for some companies this number can run upwards of ten or twenty. Rather than auditing processes or product lines piecemeal to meet specific customer requirements, global standards recognized by GFSI are benchmarked against widely-accepted, recognized food safety and quality criteria, allowing for consolidation of audits. Conducting fewer audits reduces downtime, frees up resources and greatly increases cost efficiency.
Certification to a standard is carried out by a certification body, a 3rd party that is accredited to perform food safety audits. GFSI-recognized schemes validate and monitor auditors that work for them, requiring them to meet internationally recognised accreditation rules. This provides a system of checks and balances that helps to safeguard the integrity of each GFSI audit, and helps ensure they are carried out in a consistent manner.
Food Safety Audit Structure
Audits can be classified in 3 ways based on the auditor/auditee relationship:
- First Party: This type of audit is a self-assessment, to internally verify that procedures and management strategies reflect the requirements of a standard and reflect business objectives.
- Second Party: Also known as proprietary audits, a primary organization evaluates the performance of a supplier or contractor.
- Third Party: Independent auditors from outside the organization conduct audits, often for purposes of certification.
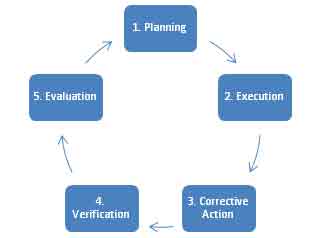
Food safety audits process, regardless of whether it is conducted by a 1st, 2nd or 3rd party, is generally comprised of five steps:
- Planning
- Execution
- Corrective and Preventive Action
- Verification
- Audit Evaluation
 Step 1 – Planning
Step 1 – Planning
Food safety audit planning should start with a clear objective. For instance, the main objective of the food safety audit may be to evaluate the management system. Or, it could focus on a specific product or product line, as requested by a customer. Other priorities may include increasing efficiency and continual improvement, and fostering a sense of ownership and participation throughout the organization.
Determining the scope of your food safety audit is another important part of planning – what areas are being targeted? This can be based on criteria such as the requirements of a standard, or upon physical boundaries such as a specific process or the organization in its entirety. Based on your objective and scope, there are three types of food safety audits:
- Focused: this type of audit targets a specific area, and may be conducted in response to a specific customer request, in response to an incident at another facility, or to conduct a pre-assessment of particular parts of the management system
- Random Sample: this type of audit lends itself mainly to spot checks or follow-up verifications of a corrective action, and provides support to problem or high-risk areas.
- Process/Department: these audits provide greater depth, and aid in the identification of trends and root cause analysis. Root cause analysis helps you to identify risks at the source, and understand how they proliferate throughout a process or facility. A deep understanding of risks requires the evaluation of all the various inter-linkages within your system and the collection and analysis of food safety audit data, and is important to achieve long-term corrective action.
Planning will also include resource and cost considerations. The food safety audit team selection is often based upon finding a balance between the resources available, the cost, and the effectiveness of the audit process. For 1st party audits, an organization may choose to use internal resources. Benefits include full availability and a steeper learning curve with regards to understanding the processes within an organization. On the other hand, external auditing resources may come already extensively trained, with up-to-date knowledge of the requirements of a standard. Some organizations may opt to use a combination of internal and external resources, share resources with similar businesses within the community, or share corporate resources amongst facilities.
Instigating a food safety audit program into an organization can bring with it the reality of outdated or non-compliant infrastructure or processes. The large costs related to updating facilities can be difficult to manage. Financing such expenditures may be facilitated by government funding, as is the case in many countries in the Americas, where money has been allocated in budgets to provide support for this purpose. In other cases, food safety businesses may need to finance the capital expenditures through more traditional means such as loans.
Not all food safety audit findings will necessarily require costly solutions. If, for example, the audits revealed a trend in which the majority of returned product was the result of human error, the initial approach may be along the lines of a weekly meeting (perhaps with complimentary coffee or food) between management and operators, where quality data is shared and the findings reported. Further monitoring of non-conformances can reveal whether a simple measure, communication, is effective, before more costly actions such as re-training or re-designing a process are implemented.
Step 2 – Execution
Audits provide a real-time assessment of the status of your operation and quality management system. More than looking at safety records and historical statistics, food safety audits look at problems that may be fermenting now – they can be proactive, rather than reactive. Looking for areas where you can implement preventative strategies in addition to reporting findings can bring value with improved efficiency and avoidance of future problems.
Food safety audits are also a good time to foster communication within the organization. Engaging with employees in interviews provides a multitude of viewpoints and is an opportunity to promote a sense of ownership and buy-in within the organization – this can go a long way in building a strong food safety culture.
Developing and using audit tools during execution, such as turtle diagrams, cross-reference matrices and process requirements matrices is very important, and will help ensure a systematic and thorough food safety audit.
Step 3 – Corrective and Preventative Action
Capturing information with proper documentation and problem descriptions provides actionable responses and timely corrective action – so it is important to develop a formal process in which responsibility is assigned and workflows clearly defined. More in-depth assessments can be afforded by root cause analysis, in which data is collected over time to identify trends, and facilitate long-term solutions rather than a “quick fix” approach. Ask yourself, what is the real issue?
Information should be communicated to everyone that is involved with the process, and they should understand their corrective responsibility. Again, an opportunity exists to enhance buy-in and ownership among employees. Detailed documentation and supporting follow-up monitoring data should be routinely captured to assess the success of corrective and preventive action.
Step 4 – Verification
This portion of the audit assesses how well corrective and preventative actions are working, and whether they are achieving their purpose as laid out in the management strategy. The person who conducts the food safety audit should not be the same person who came up with the corrective action, to add an added level of impartiality as well as a fresh viewpoint. Increasing the depth of your verification can be achieved by reviewing all of the collective outputs of a given action, as well as conducting mini-audits and follow-up interviews.
Step 5 – Audit Evaluation
Auditing your audit process in not redundant – it’s actually one of the most important parts. Are you conforming with the objectives and the food safety audit schedule, are resources being used efficiently, is there commitment on the part of management to push for implementation of corrective and preventative actions? Continual evaluation of the food safety auditing process will help ensure audits are driving value. The question should always be asked, how can we make our audits more effective? This continual loop of feedback serves to strengthen your food safety audits, and in turn your organization as a whole.
Auditing Tools
Proper reporting and management of all relevant documentation associated with food safety plans, quality management systems and auditing requires the organization and storage of large amounts of information and data. Quick and easy access to this information, as well as the tools to collect, analyse, disseminate or store it, is important.
Traditional handy auditing tools such as turtle diagrams, process requirements matrices and cross-reference matrices are augmented by new technologies, which are changing the way that audits are being conducted, from walk through to comparative analysis. Food safety and quality software are a large part of these developments, with tools designed to help meet the requirements of a given standard, customer requirement or regulator.
Food safety software can help manage documents and workflows associated with auditing and day-to-day business operations. Tasks such as the maintenance of training and certification records, reporting, data collection and analysis are stream-lined and accessible on secure intranet connections. Automating procedures imparts many benefits, by improving the efficiency and consistency of operations and simplifying many tasks.
For instance, software can enable businesses to quickly pre-assess the management system in preparation for a food safety audit, by performing a gap analysis. Each requirement of a given standard, whether it be Good Manufacturing Practices, HACCP or a GFSI standard, can be uploaded into the system along with all associated documents and workflows. A quick search can then pinpoints any gaps or non-conformances between the requirements and the system.
The monitoring of critical control points and corrective actions can also be augmented with software applications, with technologies such as hand-held devices that employees use on-the-spot to collect data and report the results into the system. Historical data is saved and easily accessible for evaluation at a later time.
Obsolete or out-of-date training materials and documents may also be managed efficiently. By automating approval processes and document control, an on-line system keep documents up-to-date, with older versions stored, and the newest one made instantly available to all designated individuals on an intranet system. Automatic notifications regarding changes to training requirements or processes also help ensure effective communication throughout the organization, and food safety requirements are continually met.
To have more articles like this emailed to your inbox, become a GFSR Member today!


